Chemical Engineering Training
Many institutes including universities provide educations on chemical engineering as the foundations to become professional process engineers. This was a good foundation in the past when the experienced engineers were available in-house to supervise the junior engineers. It was generally accepted by the company management to wait ten years for a junior engineers to become experienced in those days. However, it is no longer the case with much more competitive world today.
PreFEED provides essential principles and applications which have been proved useful in the process development, plant design and operations. We have the following concepts:
1. Practical examples
Solving practical examples is critical for acquiring the skills to handle real-world problems. We have prepared many practical examples to simulate the real work in chemical plant.
Following are the examples:
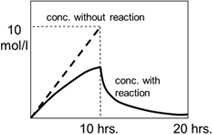
Ex1: 1st order reaction takes place by feeding A into the reactor charged with solvent. Calculate the concentration of A at 10 hours when the feed stops and at 20 hours.
Feed stars with A0=0[mol/L] and stops at 10 hours later.
k=0.1[1/hr], f=1.0[mol/L/hr]
Assume there is no dilution effect by the feed because the amount of solvent is large enough.
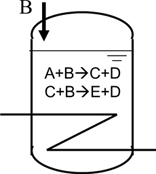
Ex2: B is fed continuously to the reactor which had been charged with A. The heat of reaction are given below.
A+B→C+D, H1=38.5 kcal/mol
C+B→E+D, H2=83.1 kcal/mol
It took 6 hours to keep the reaction temperature at laboratory scale. How long should we allow for the feeding time at the plant ?
A0 = 5.0 mol/L(others are zero at time=0)
Amount of B for feed = 2*A0( = 10 mol/L), to be added constantly
k1=8.0, k2 = 0.8[L/mol/hr]
Reaction time = feed time + 3 hour after finish of feed
Temperature = 80℃
Reactor volume = 1700L
Heat capacity of liquid = 0.6kcal/kg/K
U = 250kcal/m2/hr/k
Heat transfer area = 12m2
Cooling water = 40(in)/45℃(out)
Ex3a: 5000kg raw material is charged into empty vessel at 20℃ and is heated up to 120℃ with 140℃ steam.
Assuming no heat loss, how long does it take to heat the reactor ?

Ex3b: Half of 5000kg is charged first and the remaining half is fed to the reactor in 30 minutes. if the steam heating starts when the remaining feed begins, how long does it take to heat the reactor up to 120℃ with 140℃ steam ?
These examples are solved manually, by spreadsheet software such as Microsoft Excel, and by Equation Solving Software such as EQUATRAN.
2. Solve one problem by several ways
Solving one problem by several ways is also important for good understanding. Although process simulators can solve the complex distillation rigorously just by clicking the required information, engineers cannot gain the experience to judge if a new system is difficult or easy for separation. A simple method such as McCabe-Thiele method gives good understanding that the separation is primary dependent on the vapor-liquid equilibrium (VLE & x-y diagram). This understanding is further enhanced if the manual calculation of VLE is introduced with the learning of process simulator.
Example of Distillation Learning
McCabe-Thiele method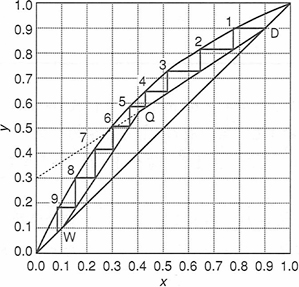
pDB-oDB VLE
- If you do not know McCabe-Thiele method, you would spend many hours using a process simulator to find p-dichlorobenzene and o-dichlorobenzene system is not appropriate for distillation.
- If you know only McCabe-Thiele method, you cannot design multi-component column, adiabatic column, azeotropic columns correctly.
- Learning distillation by several aspects give engineers with better perspectives.
3. Rules-of-thumb
While the rigorous investigation of process is required to design with minimum over-design, common senses are also valuable to make judgement in advance of detailed study and experiment. This is called Rules-of-thumb and PreFEED trainings focus on learning important Rules-of-thumb.
Example of Rule-of-thumb
Task: Size the pipe for 100 m3/hr water.

Junior engineer:
“If the velocity is 10 m/s, the size is 59 mm diameter. If it is 100 m/s, then 19mm. But I don’t know which one.”

Experienced Engineer:
“The diameter should be 130 – 190 mm, because the velocity must be 1-2 m/s for liquids.”
4. Covering wide aspects of required skills
Many skills are required to develop a process. Although several experts can contribute the projects, it is also necessary for engineers to have good understanding not only for vapor-liquid equilibrium but also reaction kinetics, solid handling and so on.
Following is the current training courses available:
| Level | Title | Duration, days | Contents |
|---|---|---|---|
| Basic Course for engineers with less than 5-year experience | Industrial Chemistry and Process Industry | 2 | Introductory course for engineers with non-chemical background |
| Essences of Practical Chemical Engineering | 4 | Overview of unit operations with plant examples | |
| Data Analysis and Introduction to Modeling | 4 | Analysis of plant data and introduction to modeling | |
| Intermediate Course for senior engineers | Phase Equilibrium and Its Applications | 2 | Understanding of principles of phase equilibrium and application in activity coefficient models |
| Development of Practical Modeling Skills | 3 | Development of modeling skills for major unit operations such as extraction, evaporation, drying and so on | |
| Reaction Engineering and Its Applications | 5 | development of modeling and analysis skills for various reaction processes | |
| Process Simulator 1: physical properties | 2 | Training by AspenPlus | |
| Process Simulator 2: vapor-liquid equilibrium and basic distillation | 3 | Training by AspenPlus | |
| Process Simulator 3: advanced distillation | 3 | Training by AspenPlus | |
| Comparison of batch and continuous operations | 3 | Understanding the differences between batch and continuous operations for major unit operations | |
| Advanced Course | Workshop on Process Development | 10 | Learn the skill to develop a whole process from the experimental data (for a specialty chemical and a petrochemical processes) |
We are happy to customize our courses to your needs. Please contact us by email marketing@prefeed.com.