EQUATRAN on the web - Try Our Examples
Ex1a: Determination of NRTL Parameters
problem description
Determine NRTL parameters for the isobaric data of ethanol-water system.
solving principles
Optimize NRTL parameters (T12, T21) in the following equations by minimizing the error.
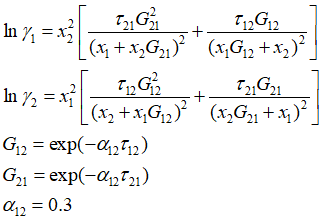
The error is defined as relative errors of activity coefficients.
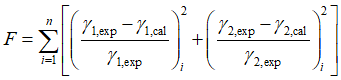
results

tips to tailor to your problems
If you want to change the components, do the following:
[ Change Antoine's parameters ]
![]()
[ Change number of data points ]
![]()
[ Change VLE data set ]
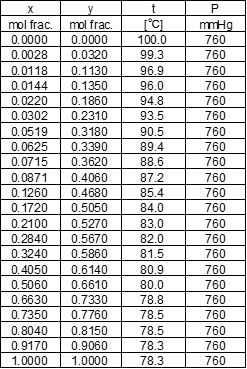
ethanol liquid mol fraction, ethanol vapor mol fraction, temperature, total pressure:

Ex1b: Calculation of VLE using NRTL parameters
problem description
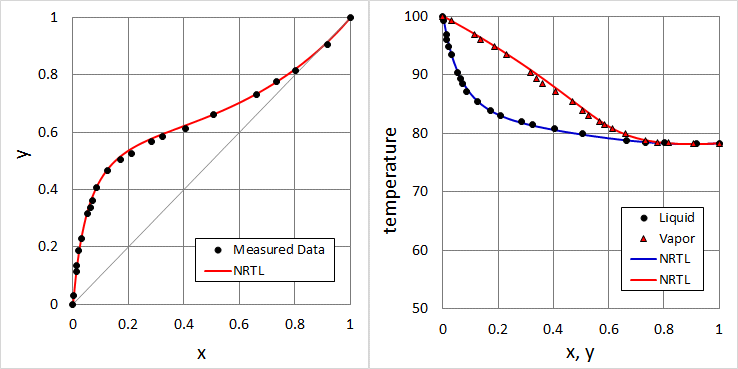
Calculation of VLE to compare with measured data using NRTL parameters determined in the previous example problem.
results
Ex2: Determination of Reaction Kinetic Constants
problem description
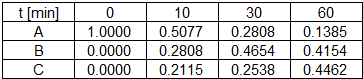
Determine reaction kinetic constants, k1 and k2 for the reaction data below.
![]()
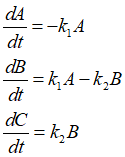
solving principles
Assume k1 and k2 to calculate trend of concentrations of A, B and C and calculate errors with measured data. Optimize k1 and k2 by Marquardt method, non-linear dynamic optimization.
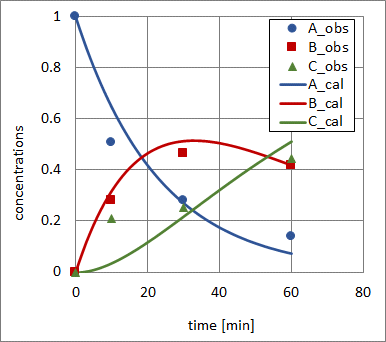
results

tips to tailor to your problems
If you want to change the reaction equations, which are three-component first order series reactions, modification of the codes is not transparent, contact PreFEED for assistance.
Please include your affiliates (company, telephone, and name) in the email.
Other examples of coding the equations
The following sample problems will help you understand how EQUATRAN-G works.
Lesson 1 : Antoine Equation
Lesson 2 : Heat Balance of Heat Exchanger
Lesson 3 : Boiling Point Calculation
Lesson 4 : Reaction Calculation
Lesson 5 : Simple Distillation